 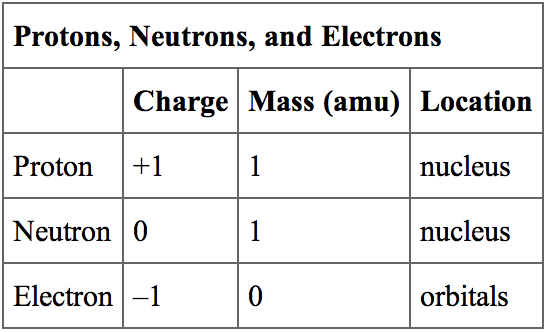
These are referred to as the elementary charge. This is, appropriately, referred to as "confinement." This is why we don't observe quarks-and therefore their fractional charges-outside their composite particles (such as protons and neutrons). A proton has a charge of +1.602 1019 and an electron has a charge of 1.602 1019. Quarks are confined to the particles they compose. Likewise, neutrons are composed of two down quarks and one up quark, so the total charge is 0. This fundamental property of matter is known as electric charge. 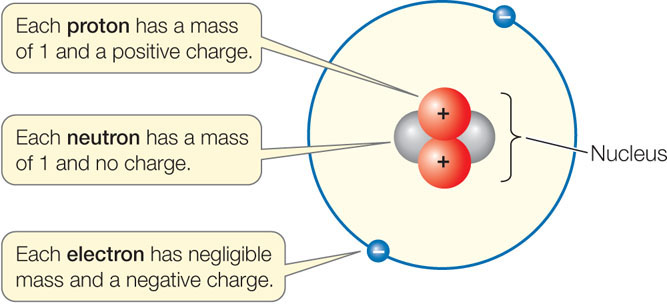
Protons are composed of two up quarks and one down quark, so the total charge is +1. This means that electrons have a negative charge (-1) while protons have a positive charge (+1). A QUICK REVIEW OF OLD STUFF Electrons and protons have the same magnitude (amount) of charge, although the electron is negative and the proton positive. Up, charm, and top all have fractional charge of +2/3, while down, strange, and bottom all have a charge of -1/3. Two of the subatomic particles have electrical charges: protons have a positive charge while electrons have a negative charge. A neutron, like the name implies, is neutral with no net charge. Solution Verified by Toppr Correct option is D) Given : m h1.67×10 27 kg Net charge on hydrogen atom q(e+e)ee Let the distance between them be d. It turns out that protons and neutrons are composed of particles called "quarks." These quarks, which come in different "flavors" (up, down, charm, strange, top, bottom) make up certain particles. A proton has positive charge of 1, that is, equal but opposite to the charge of an electron. However, there are particles with fractional charge. The potential energy of the electron in the field of the positive proton point charge is U(r) -qeV(r) - keqe2/r. 
Therefore, the charges of such particles are integer values. Each electron has a charge equal in magnitude to that of a proton. Both electron and proton are the fundamental components of an atomic structure and has. The charge of an atom or composite particle is found by adding the charges of its protons and electrons (since neutrons are electrically neutral). Charges on electron and proton are given below:- Charge on electron -1.6 x 10 -19 coulomb it is absolute charge of electron. A given element can have a variable number of neutrons thus, while atoms of the. As against, a proton is a charged particle having a positive charge. So, protons have charge +1, and electrons, -1, using units of e. In the SI system of units, the value of the elementary charge is exactly defined as e īy combining the best measured value of the antiproton charge (below) with the low limit placed on antihydrogen's net charge by the ALPHA Collaboration at CERN.Usually, if charge is described as just a number, it is in units of e, or the charge of a proton, which is approximately 1.6x10 -19 Coulombs (also the absolute value of the charge of an electron). The periodic table is a chart that organizes elements by their atomic structure. This elementary charge is a fundamental physical constant. The elementary charge, usually denoted by e, is the electric charge carried by a single proton or, equivalently, the magnitude of the negative electric charge carried by a single electron, which has charge −1 e. 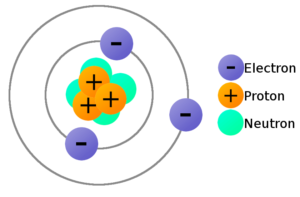
Charge carried by one proton or electron Elementary charge 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
